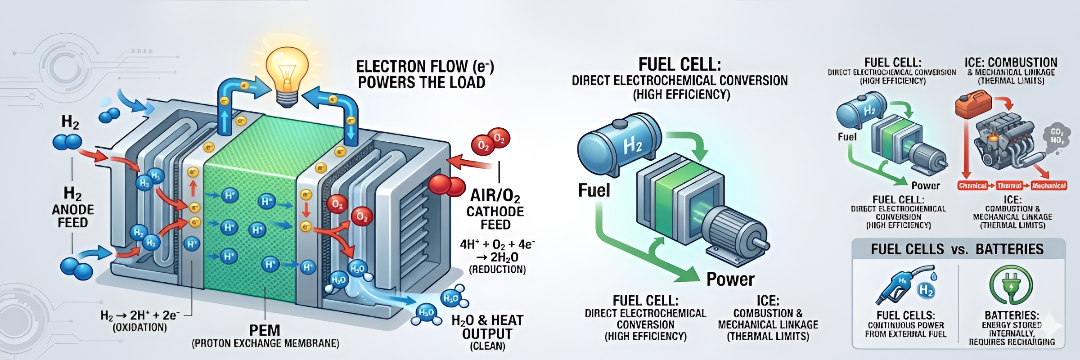
A fuel cell is a sophisticated energy conversion device that transforms the chemical energy of a fuel directly into electricity. Unlike traditional combustion engines, this process bypasses intermediate thermal or mechanical stages, offering a cleaner and more efficient power solution.
The Principle of Operation: Electrochemical vs. Combustive
While both fuel cells and internal combustion engines (ICE) release energy by reacting fuel with oxygen, the methods are fundamentally different:
Direct Conversion: The electrical energy generated by a fuel cell can be used for work directly, while the byproduct heat is either utilized for other purposes or safely expelled.
Internal Combustion Engines: React fuel and oxidant combustively, releasing energy as heat to move a piston.
Fuel Cells: React fuel and oxidant electrochemically, releasing energy as low-voltage DC electricity and heat.
Anatomy of a Galvanic Cell
At its heart, every fuel cell is a galvanic cell, a category it shares with standard batteries. These cells consist of three essential components:
- The Anode (Negative Electrode): A substance that readily releases electrons through oxidation. In a fuel cell, this is typically hydrogen gas.
- The Cathode (Positive Electrode): A substance that readily accepts electrons through reduction. In a fuel cell, this is typically oxygen or air.
- The Electrolyte: A specialized material that conducts ions but acts as an electrical insulator, preventing a short circuit between the electrodes.
The Electrochemical Reaction
The magic of a fuel cell happens through specific chemical reactions at each electrode. In a standard hydrogen fuel cell, the process is governed by these equations:
The reactions at the anode are:
H2 ➡ 2H+ + 2e–
The reaction at the cathode is:
½O2 + 2e– + 2H+ ➡ H2O
The H+ ion is drawn through the electrolyte from the anode to the cathode by the reactive attraction of hydrogen to oxy-gen, while electrons are forced through an external circuit. Combining the anode and cathode reactions,
The overall cell reaction is:
H2 + ½O2 ➡ H2O
Thus, the fuel cell produces water that accumulates at the cathode. This product water must be continually removed to facilitate further reaction.
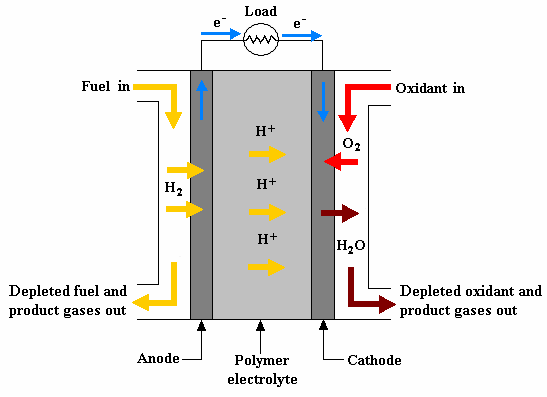
PEM fuel cell illustration
The Journey of an Electron: A Molecular Perspective
At the Anode: The Split (H2 ➡ 2H+ + 2e–)
- Fuel Feed: Hydrogen gas (H2) is supplied to the anode.
- Ion Migration: Fuel atoms dissolve into the electrolyte to become positively charged hydrogen ions (H+ ions).
- Electron Release: This process leaves behind free electrons (e–), creating a negative charge buildup within the anode
The Electrolyte Barrier: Strict Insulation
- The Filter: The electrolyte conducts ions (H+) but acts as an electrical insulator.
- The Trap: Because the electrolyte cannot conduct electricity, the electrons (e–) remain trapped at the anode and cannot pass directly to the cathode.
The External Load: Doing Useful Work
- External Flow: To complete the reaction, these e- are forced to flow through an external circuit toward the cathode.
- Powering the Motor: On this journey, the e- pass through an external load, performing useful work such as powering an electric motor.
At the Cathode: Completing the Reaction (½O2 + 2e– + 2H+ ➡ H2O)
- Oxidant Feed: Oxygen or air (O2) is supplied to the cathode.
- Reunion: Once the electrons (e–) arrive via the external circuit, they join the H+ ions that migrated through the electrolyte and the O2 atoms.
- Final Product: This reunion forms the final reaction product: pure water (H2O) and heat.
Demand-Driven Power
- Load Control: The entire rate of this electrochemical reaction—and thus the consumption of H2 and O2 is determined entirely by the size of the electrical load applied to the system.
Fuel Cells vs. Batteries vs. ICE
Understanding how these technologies compare is crucial for identifying their best applications:
Fuel Cells vs. Batteries
Reactant Storage: Batteries store energy internally as part of their physical structure and are consumed during use. Fuel cells use reactants supplied from an external source and do not need recharging as long as fuel is available.
Electrode Material: Battery electrodes are typically solid metals like zinc or lithium. Fuel cell electrodes are gases (Hydrogen and Oxygen) often in contact with a platinum catalyst.
Fuel Cells vs. Internal Combustion Engines (ICE)
Form Factor: Both systems draw gaseous, hydrogen-rich fuel from external storage.
Operation: ICEs are mechanical devices that generate mechanical energy, while fuel cells are solid-state devices that generate electrical energy.
Emissions: Fuel cells operating on pure hydrogen produce zero harmful emissions. Standard ICEs produce significantly more pollution unless specifically configured for pure hydrogen.
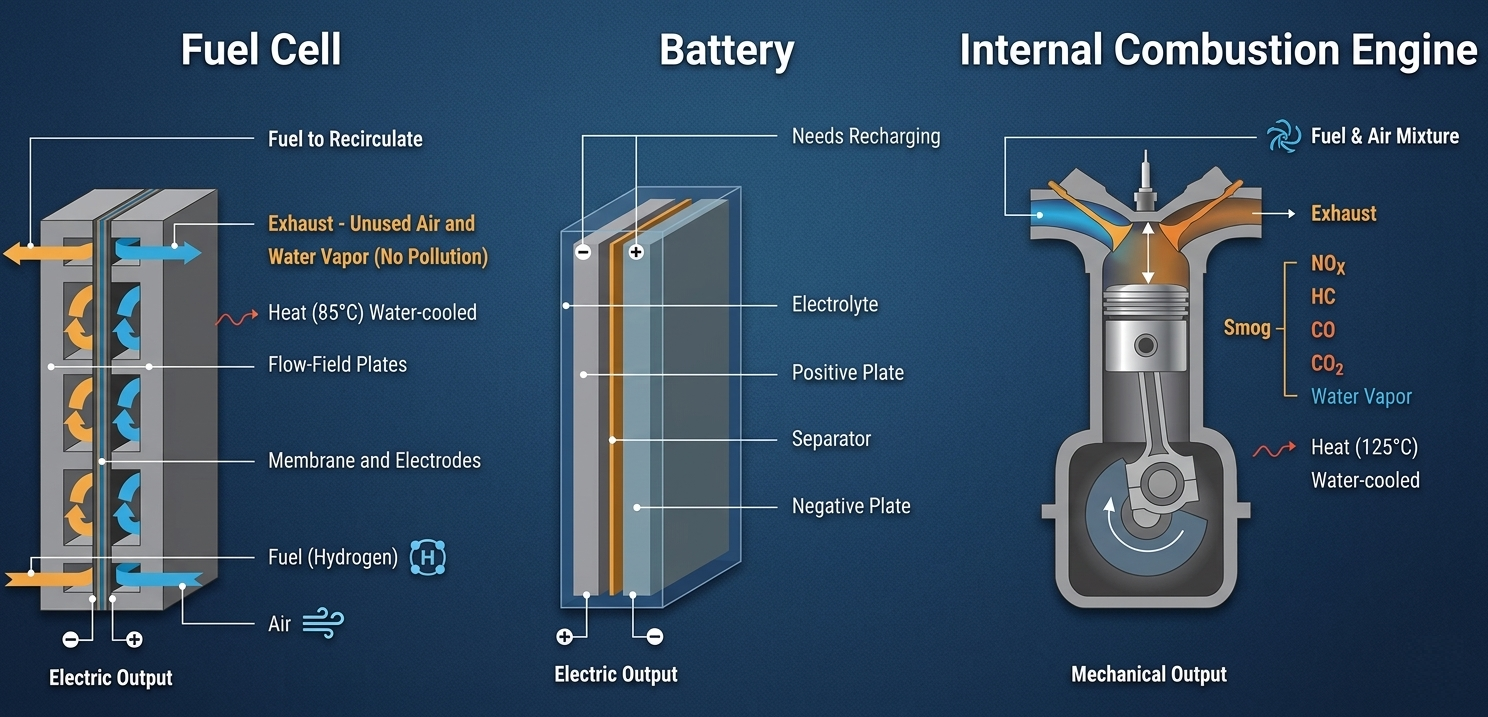
Comparison between Fuel Cell, Battery and Internal Combustion Engine
Technical Characteristics Comparison
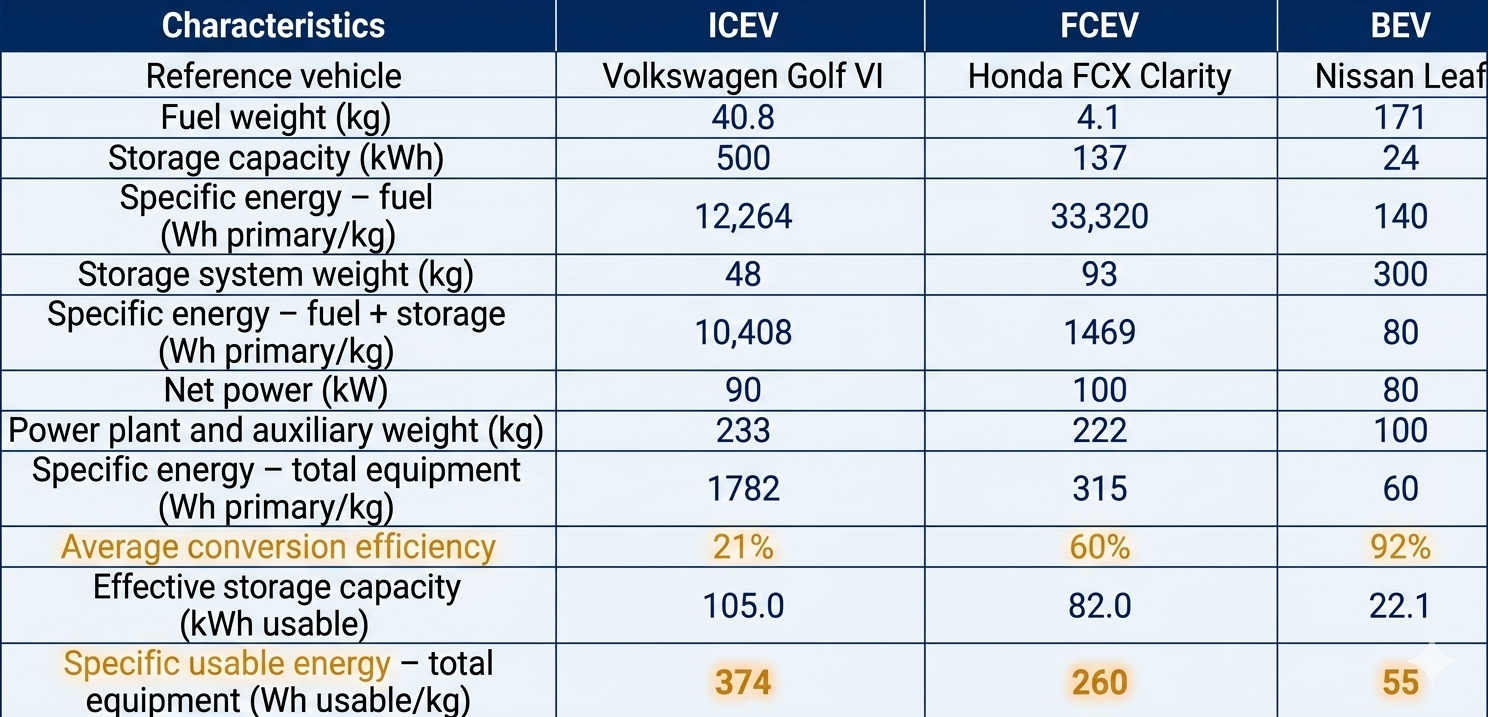
Characteristics comparison of internal combustion engine vehicles (ICEVs), fuel cell electric vehicles (FCEVs) and battery electric vehicles (BEVs)