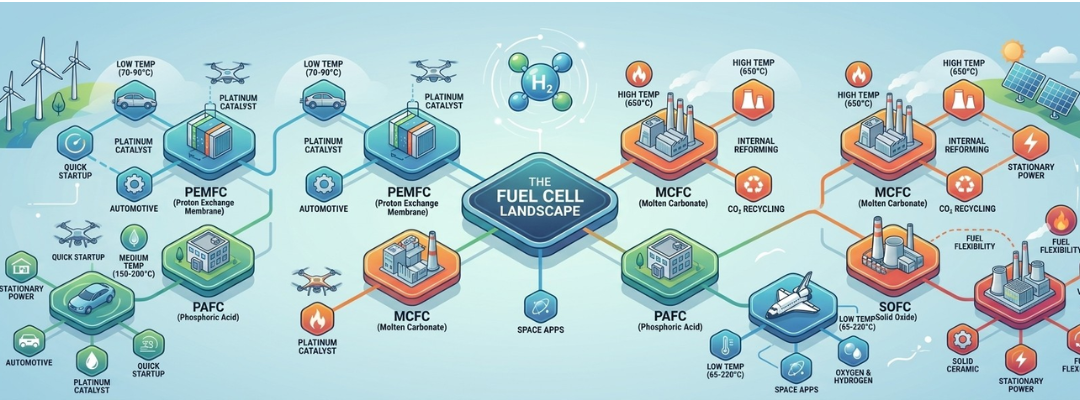
Types of Fuel Cells
Fuel cells are electrochemical devices that convert chemical energy from a fuel directly into electrical energy. While they all share this fundamental purpose, they differ significantly in their design and application. The primary distinction among fuel cell types is the electrolyte they employ.
This choice of electrolyte is crucial because it directly dictates the fuel cell’s operating temperature, which in turn influences its efficiency, startup time, material requirements, and suitability for different uses. Fuel cells are generally categorized into two main groups based on this temperature: high-temperature and low-temperature.
Categorization based on Operating Temperature
High-Temperature Fuel Cells
These fuel cells operate at temperatures exceeding 1100 ºF (600 °C). Their high-temperature operation offers several distinct advantages, including:
- Spontaneous Internal Reforming: They can internally and spontaneously reform light hydrocarbon fuels (like methane) into hydrogen and carbon in the presence of water. This steam reforming process occurs at the anode over a nickel catalyst, provided sufficient heat is available.
- Fuel Flexibility: Internal reforming eliminates the need for a separate fuel processor and allows the use of fuels other than pure hydrogen. This can lead to an overall efficiency increase of as much as 15%.
- High-Grade Waste Heat: They generate high-grade waste heat suitable for downstream co-generation processes.
- Inexpensive Catalysts: High-temperature fuel cells can react easily and efficiently without the need for expensive noble metal catalysts like platinum.
- However, high-temperature operation also presents significant challenges:
- Materials Problems: Severe materials problems arise as few materials can withstand a chemical environment at high temperatures for extended periods without degradation.
- Slow Startup and Scale: They are not suitable for quick startup and do not easily lend themselves to large-scale operations.
- Reduced Energy Release: The amount of energy released by the electrochemical reaction actually degrades as the reaction temperature increases.
Due to these factors, current applications for high-temperature fuel cells are primarily focused on stationary power plants, where the benefits of internal reforming and co-generation capabilities outweigh the disadvantages of material breakdown and slow startup.
Prominent high-temperature fuel cell types
- Molten Carbonate
- Solid Oxide
Low-Temperature Fuel Cells
Low-temperature fuel cells typically operate below 480 ºF (250 ºC). Their key characteristics are:
- External Hydrogen Requirement: Their low operating temperatures do not permit internal reforming, so they require an external source of hydrogen.
- Quick Startup and Vehicle Suitability: They exhibit quick startup, suffer fewer materials problems, and are easier to handle for vehicle applications.
Prominent low-temperature fuel cell types
- Alkaline
- Phosphoric Acid
- Proton Exchange Membrane (or solid polymer)

Comparison of different fuel cell technologies