The primary difference between these two technologies lies in the operating temperature and the mechanics of proton conduction. While both use a solid polymer electrolyte, the thermal environment changes the chemical requirements of the system entirely.
1. Low-Temperature PEMFC (LT-PEMFC)
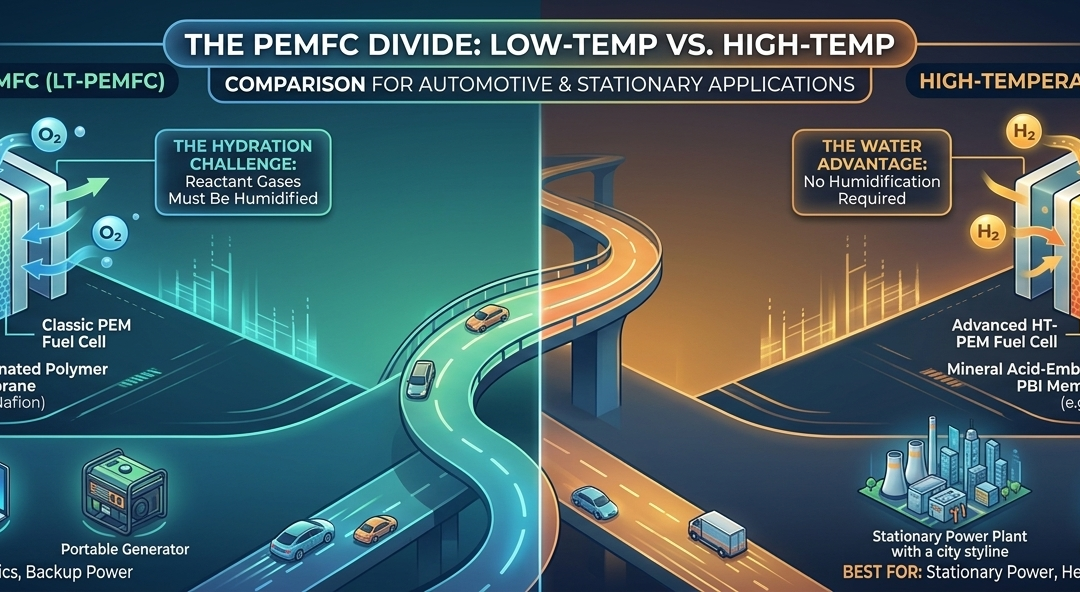
This is the “classic” PEM fuel cell. It typically operates between 140 °F and 176 °F (60 °C to 80 °C).
- The Membrane: It uses a fluorinated polymer (like Nafion) that requires liquid water to conduct protons.
- The Hydration Challenge: Because it relies on water, the reactant gases must be humidified. If the cell gets too hot (near 212 °F/100 °C), the water evaporates, the membrane dries out, and proton conductivity stops.
- Startup & Safety: LT-PEMFCs excel in rapid startup and are considered very safe due to their low-pressure and low-heat operation.
- Best For: Fuel cell vehicles (FCEVs), portable electronics, and small-scale backup power.
2. High-Temperature PEMFC (HT-PEMFC)
HT-PEMFCs push the boundaries, operating between 250 °F and 390 °F (120 °C to 200 °C).
- The Membrane: Instead of water-reliant Nafion, these use mineral acids (like phosphoric acid) embedded in a high-performance polymer (like Polybenzimidazole or PBI).
- The Water Advantage: Because they operate above the boiling point of water, they do not require humidification systems. This significantly simplifies the mechanical design and reduces the weight of the overall system.
- Carbon Monoxide (CO) Tolerance: As noted in your reference file, CO is a “poison” to fuel cells. However, at higher temperatures, the catalyst becomes much more resistant to CO, allowing HT-PEMFCs to use lower-quality hydrogen (reformate) without failing.
- Best For: Stationary power plants, heavy-duty shipping, and aviation where high-grade waste heat can be recycled for extra efficiency.
Comparative Analysis: Which One Wins?